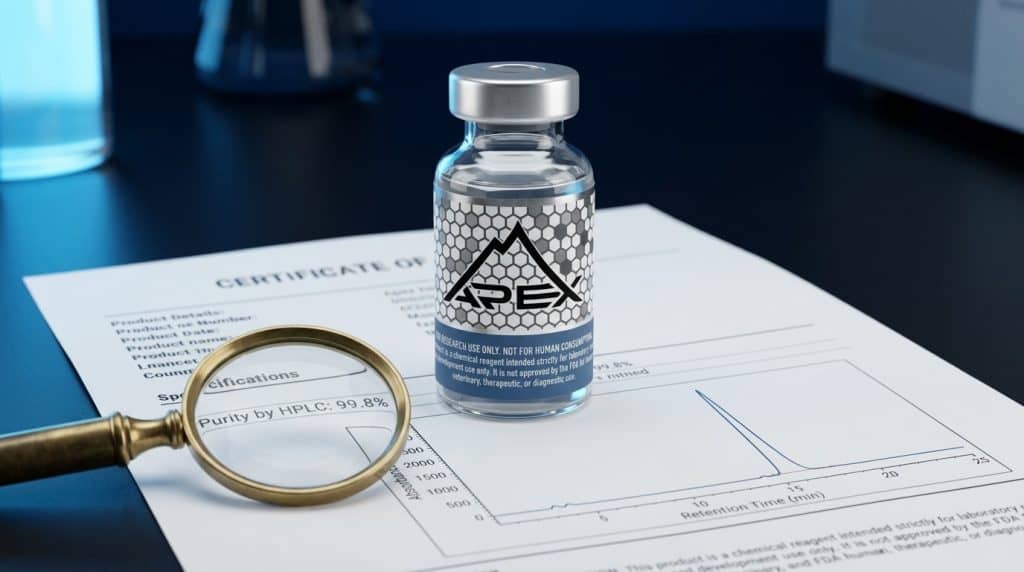
A Certificate of Analysis (COA) is the single most important document in peptide quality verification. It is the analytical report that tells you whether the compound in your vial is actually what the label says it is, at the purity the supplier claims. Yet despite its importance, many researchers do not know how to properly read, interpret, or critically evaluate a COA — which is exactly what low-quality suppliers rely on. Understanding what a legitimate COA looks like, what each section means, and what red flags to watch for is essential for protecting the integrity of your research data.
This guide teaches you how to read every section of a peptide COA, understand the analytical methods used to generate the data, and evaluate whether a COA is trustworthy. At Apex Laboratory, our dual HPLC and Mass Spectrometry verification process generates the analytical data that forms the basis of every COA we provide.
What Is a Certificate of Analysis?
A Certificate of Analysis is an analytical document provided by a peptide manufacturer or supplier that reports the results of quality control testing performed on a specific batch of product. A proper COA should accompany every batch of research peptides you purchase and should contain, at minimum, the following elements: the compound name and chemical identity, the batch or lot number, the date of analysis, the analytical methods used (HPLC, Mass Spectrometry, etc.), the results of each test, and the identity of the laboratory that performed the analysis.
The COA is not a marketing document — it is a factual report of measured analytical data. A legitimate COA presents objective numbers that either confirm or fail to confirm the product’s identity and purity. It should be specific to the batch you received, not a generic template reused across all batches.
The Two Core Tests on Every Peptide COA
1. HPLC (High-Performance Liquid Chromatography) — Purity Testing
HPLC is the gold standard analytical method for determining peptide purity. It works by dissolving the peptide sample in a mobile phase solvent and passing it through a column packed with stationary phase particles. Different molecular species travel through the column at different rates based on their chemical properties — the main peptide separates from any impurities, degradation products, or synthesis byproducts. A UV detector at the end of the column measures the absorbance of each component as it elutes, generating a graph called a chromatogram.
On a chromatogram, each distinct peak represents a different molecular species in the sample. The main peptide should appear as a single, sharp, dominant peak. Impurities appear as smaller peaks at different retention times. The purity percentage is calculated by comparing the area of the main peak to the total area of all peaks in the chromatogram. For example, if the main peak accounts for 99.2% of the total peak area, the HPLC purity is reported as 99.2%. For a deeper explanation of how HPLC technology works, see our companion article: Understanding HPLC Testing for Peptide Purity.
What to Look for on the HPLC Section of a COA
- Purity percentage: For research-grade peptides, look for ≥95% minimum, with ≥98% being good and ≥99% being excellent. All Apex Laboratory peptides are verified to ≥99% purity.
- A chromatogram image: A legitimate COA should include the actual chromatogram graph — not just a number. The chromatogram lets you visually confirm that the main peak is clean, sharp, and dominant, with no significant secondary peaks.
- Retention time: The time at which the main peak elutes from the column. This should be consistent with the expected retention time for the specific peptide being tested.
- Method details: Column type, mobile phase composition, flow rate, and detection wavelength. Reputable labs report these so the analysis can be independently reproduced if needed.
2. Mass Spectrometry (MS) — Identity Verification
While HPLC tells you how pure the sample is, Mass Spectrometry tells you what the sample actually is. MS works by ionizing the peptide molecules and measuring their mass-to-charge ratio (m/z), which reveals the molecular weight. If the measured molecular weight matches the expected molecular weight of the target peptide, the compound’s identity is confirmed.
This is critically important because HPLC alone cannot tell you whether a pure sample is the correct compound — it can only tell you that the sample contains predominantly one substance. A sample could be 99.5% pure by HPLC but be the wrong peptide entirely. Mass Spectrometry is what closes this gap by confirming molecular identity. This is why Apex Laboratory uses both HPLC and Mass Spectrometry on every batch — purity without identity confirmation is incomplete quality assurance.
What to Look for on the Mass Spec Section of a COA
- Observed molecular weight: This should match the expected (theoretical) molecular weight of the peptide within an acceptable margin. For most peptides, the observed mass should be within ±1 Da (dalton) of the expected mass.
- Expected molecular weight: The theoretical MW should be listed for comparison. For example, BPC-157 has an expected MW of 1419.53 g/mol — the observed mass should be very close to this value.
- A mass spectrum image: The actual spectrum showing the m/z peaks. The dominant peak should correspond to the target peptide’s mass.
- Ionization method: Common methods include ESI (Electrospray Ionization) and MALDI (Matrix-Assisted Laser Desorption/Ionization). ESI is more common for peptides in the 500-10,000 Da range.
Apex Laboratory Quality Process
Every batch undergoes dual HPLC + Mass Spectrometry verification to ≥99% purity before entering inventory. Learn about our process · Request COA documentation
Additional Tests You May See on a COA
Some COAs include additional testing beyond HPLC and Mass Spec. These are not always present but add further quality assurance when included:
Amino Acid Analysis (AAA)
This test hydrolyzes the peptide into its individual amino acids and quantifies each one. The measured amino acid ratios are compared to the expected ratios based on the peptide’s known sequence. This confirms not just molecular weight (which Mass Spec provides) but the actual amino acid composition.
Endotoxin Testing (LAL Test)
The Limulus Amebocyte Lysate (LAL) test detects bacterial endotoxins. This is particularly relevant for peptides that will be used in cell culture or biological assays where endotoxin contamination could interfere with results. A negative or low endotoxin result (typically reported as <0.5 EU/mg) is desirable.
Peptide Content / Net Peptide Weight
This reports the actual peptide content as a percentage of the total weight in the vial. The vial weight includes not only the peptide itself but also counter-ions (acetate, TFA), residual moisture, and salts. A typical peptide content is 60-85% by weight. This means a vial labeled “5 mg” might contain 5 mg total weight but only ~3.5-4 mg of actual peptide. High-quality suppliers account for this and may adjust fill weights accordingly.
Appearance and Solubility
Basic physical characterization — the COA may note the powder appearance (white to off-white lyophilized powder) and solubility in water or other solvents. These are simple quality checks that confirm the product matches expected physical properties.
Red Flags: How to Spot a Fake or Low-Quality COA
Unfortunately, not all COAs in the peptide industry are legitimate. Some suppliers provide fabricated, recycled, or misleading documentation. Here are the red flags to watch for:
- No chromatogram or mass spectrum image included. A COA that reports a purity percentage but doesn’t show the actual chromatogram is immediately suspect. The graph is the evidence — without it, you’re taking the supplier’s word on faith.
- No batch or lot number. Every legitimate COA should be traceable to a specific manufacturing batch. A COA without a lot number may be a generic template reused across all products.
- Identical COAs across different products or batches. If you purchase two different peptides (or the same peptide at different times) and the COAs have identical chromatograms, retention times, and mass spectra, they are almost certainly fabricated. Each batch should produce unique analytical data with slight natural variations.
- Suspiciously round purity numbers. Purity of exactly “99.00%” is less credible than “99.24%” or “98.7%.” Real analytical measurements produce specific, non-round numbers. Perfect round numbers often indicate the value was chosen rather than measured.
- No testing laboratory identified. A legitimate COA should state which laboratory performed the analysis — either the manufacturer’s in-house QC lab or a named third-party testing facility.
- No analytical method details. If the COA doesn’t mention the column type, mobile phase, or any experimental parameters, it may not be a real analytical report.
- Missing date of analysis. Without a date, you cannot confirm whether the testing was performed on the current batch or is recycled from a previous one.
How to Request and Verify COA Documentation
When purchasing research peptides from any supplier, including Apex Laboratory, follow these best practices for COA documentation:
- Request the COA before purchasing if your internal quality requirements mandate pre-purchase documentation review. Most reputable suppliers will provide COAs upon request.
- Match the lot number on the COA to the lot number printed on the vial you receive. They should correspond to the same batch.
- Verify the molecular weight on the Mass Spec report matches the known molecular weight of the compound you ordered. For example, if you ordered Semaglutide (expected MW: 4113.58 g/mol), the observed mass should be within ±1 Da of that value.
- If in doubt, run your own independent analysis. The ultimate verification is third-party testing at an independent analytical laboratory. You can submit a small sample from your received vial for independent HPLC and Mass Spec analysis. If the supplier’s COA is legitimate, the independent results will closely match.
Frequently Asked Questions
Should every peptide purchase come with a COA?
Ideally, yes. Any reputable research peptide supplier should be able to provide batch-specific analytical documentation for the products they sell. At Apex Laboratory, COA documentation is available upon request for all current batches. If a supplier cannot or will not provide analytical documentation, that is a significant red flag.
What is the difference between HPLC purity and Mass Spec purity?
These terms refer to different measurements. HPLC purity measures chromatographic purity — the percentage of the sample that is the desired compound versus impurities, based on peak area comparison. Mass Spectrometry doesn’t measure “purity” in the same way — instead, it confirms molecular identity by measuring the compound’s molecular weight. Together, they answer two complementary questions: “How pure is it?” (HPLC) and “Is it the right compound?” (Mass Spec). Both are needed for complete quality verification.
What purity percentage should I look for when buying research peptides?
For most research applications, ≥98% HPLC purity is considered good quality, and ≥99% is excellent. Purity below 95% may contain enough impurities to affect experimental results, particularly in sensitive biological assays. All peptides from Apex Laboratory are verified to ≥99% purity through dual HPLC and Mass Spectrometry analysis.
Can I trust a COA from a third-party lab more than one from the manufacturer?
Third-party COAs from independent, accredited analytical laboratories are generally considered the most trustworthy because they have no financial incentive to report favorable results. In-house manufacturer COAs can be equally valid if the manufacturer has a rigorous quality system, but independent third-party verification adds an additional layer of assurance. When in doubt, request third-party documentation or submit a sample for your own independent testing.
What does TFA content on a COA mean?
TFA (trifluoroacetic acid) is a counter-ion commonly used during peptide purification via reverse-phase HPLC. TFA salts often remain associated with the peptide after purification and contribute to the total vial weight. TFA content is typically reported as a percentage (often 5-15% of total weight) and is factored into the “net peptide content” calculation. TFA itself is generally considered inert at the trace levels present in research peptide preparations, though some sensitive cell-based assays may require TFA-free (acetate salt) formulations.
Continue Your Research
- Understanding HPLC Testing: How Peptide Purity Is Verified
- How to Reconstitute Peptides: A Complete Step-by-Step Lab Protocol
- Peptide Storage Guide: Temperature, Stability & Shelf Life
- Peptide Reconstitution Calculator — Free Research Tool
Research Use Disclaimer
This article is provided for educational and research reference purposes only. All peptides sold by Apex Laboratory are intended exclusively for in-vitro laboratory research use and are not for human consumption. Contact our support team at support@apexlaboratory.org to request COA documentation for any specific product or batch.